|
An example is neon, which has the element symbol Ne, atomic number 10 and mass. The electron configuration of silicon is 1s2 2s2 2p6 3s2 3p2. The symbol for the element is as shown below: AZE (1) (1) Z A E. The average silicon atom has fourteen protons, fourteen electrons, and most have 14 neutrons. We can determine the number of neutrons as #14-6=8# neutrons.Įxample 2. Every nuclide has a chemical element symbol (E) as well as an atomic number (Z), the number of protons in the nucleus, and a mass number (A), the total number of protons and neutrons in the nucleus. Now write the isotopic notation for carbon-14. The name carbon-14 tells us that this isotope's mass number is #14#. Isotope notation, also known as nuclear notation, is important because it allows us to use a visual symbol to easily determine an isotope's mass number, atomic number, and to determine the number of neutrons and protons in the nucleus without having to use a lot of words.Įxample 1: What is the isotopic notation for the isotope carbon-14?įrom the periodic table, we see that the atomic number (number of protons) for the element carbon is #6#. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. 
However, because different isotopes have different numbers of neutrons, they can differ in mass number, which is the sum of the protons and neutrons in the nucleus. Nitrogen is a chemical element with atomic number 7 which means there are 7 protons in its nucleus. All atoms of the same element have the same number of protons, which is the atomic number of that element. 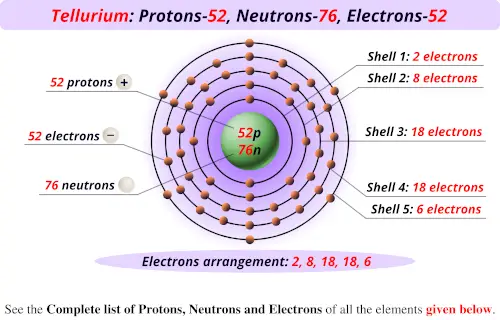
Isotopes are atoms of the same element that differ in the number of neutrons in their atomic nuclei.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
